Tandem catalysis and smart design offer new route to polypropylene plastic | Research
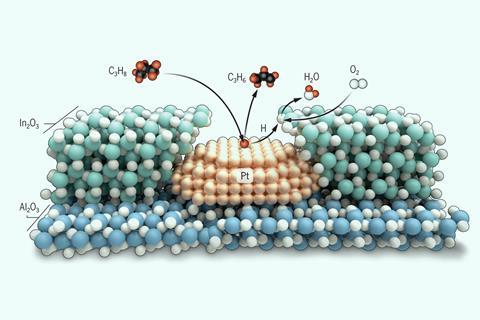
Two catalysts were combined in such a way that the production of a key plastic could be revolutionized. The Northwestern University team’s approach uses platinum nanoparticles to dehydrate propane into propylene, a plastic precursor, and selectively burns the resulting hydrogen with an indium (III) oxide coating.
“This major advance is the production of a material that can perform the dehydrogenation step and the selective combustion step simultaneously and under the same conditions,” says Justin Notestein of Northwestern. The team achieved yields of 37% percent, compared to a previous high from other researchers of 20%. “I didn’t think it would work that well,” Notestein admits.
Polypropylene is becoming increasingly popular because it is a relatively easy-to-recycle plastic. As a result, demand for its precursor, propylene, is likely to exceed supply, according to Notestein. The production of propylene by the oxidative dehydrogenation of propane has been proposed, but “without sufficient success to warrant industrial application,” he adds. Notestone’s group had already been working on the problem with the team of his northwestern colleague Peter Stair and wanted to try again.
However, the researchers – especially the postdoctoral fellow Huan Yan – were faced with what Notestein describes as an “annoying challenge” when dealing with hydrogen. The dehydrogenation of propane is an equilibrium process, which means that building up the hydrogen by-product slows the process down. The platinum catalyst required for propane dehydration can burn hydrogen and react with oxygen to form water. However, this is not selective and there is a risk that valuable propane and propylene will be burned at the same time. “Poorly selective propane oxidation isn’t particularly interesting unless you’re trying to grill a hamburger in your yard,” says Notestein.
Other scientists have avoided such challenges by separating the dehydrogenation and combustion steps either sequentially or in different parts of a reactor. The Northwestern researchers took inspiration from larger-scale strategies in which selective hydrogen combustion catalysts are placed so that they consume all of the available oxygen before the reaction mixture hits the dehydrogenation catalyst.
Cleaner plastics
The team therefore wanted to replicate this on the nano-scale and apply an outer layer of indium (III) oxide over an inner core of a platinum nanoparticle catalyst supported on aluminum oxide. Yan used a technique called Atomic Layer Deposition (ALD), which is commonly used in nanofabrication but has only recently become available in this catalysis context. Notestein says the main advantage of ALD is that it “allows us to organize catalytic functions perpendicular to a surface rather than just parallel”.
Student Macy Nanda and PhD student Izabela Samek developed the ALD protocol, and student Dian Jing built a kinetic reaction model that “was very helpful in showing us how to think about chemistry,” says Notestein. “There is evidence that we transfer hydrogen atoms directly from the platinum surfaces to the indium oxide by surface diffusion,” he says. “We believe this is critical in order to use up hydrogen atoms and oxygen very quickly.”
Giulia Tarantino of Imperial College London, UK, says this is “a great example of how tandem catalysis coupled with intelligent catalyst design can address the current challenges in chemical manufacturing”. “A really important element of this work is the coupling between propane dehydrogenation and selective hydrogen combustion, which greatly increases propylene selectivity,” she says.
Jinlong Gong from Tianjin University in China calls this work “elegant” and “an intelligent design of tandem catalysis”. He adds, however, that “the technology to control the structures of this catalytic system at the atomic level needs to be developed on an industrial scale”.
This study showed that ALD “is a very effective way to modify catalysts,” says Notestein. Companies are now working to broaden the approach. ALD also enables a general catalytic strategy to get active oxide onto a metal, he says. His team is now trying to apply the approach to other tandem reactions of light alkanes.


Comments are closed.